January 21, 2022
Periodontal dipeptidyl peptidase 7 has a wide range of substrate properties and is involved in blood glucose regulation through this activity
Expanded substrate specificity supported by P1’ and P2’ residues enables bacterial dipeptidyl-peptidase 7 to degrade bioactive peptides
Yuko Ohara Nemoto1, Yu Shimoyama2, Toshio Ono1, Mohammad Tanvir Sarwar1,4, Manami Nakasato3, Minoru Sasaki2, and Takayuki K. Nemoto1*
1Department of Oral Molecular Biology, Course of Medical and Dental Sciences, Nagasaki University Graduate School of Biomedical Sciences, 1-7-1 Sakamoto, Nagasaki 852-8588, Japan 2Division of Molecular Microbiology, Department of Microbiology, Iwate Medical University, 1-1-1 Idai-dori, Yahaba-cho, Iwate 028-3694, Japan
*Corresponding author
https://www.jbc.org/article/S0021-9258(22)00025-4/fulltext
Professor Emeritus Takayuki Nemoto and visiting researcher Yuko Ohara-Nemoto at Nagasaki University School of Dentistry, in collaboration with Professor Minoru Sasaki and Associate Professor Yu Shimoyama at Iwate Medical University, have shown for the first time that dipeptidyl peptidase (DPP) 7 of periodontal bacteria has broad substrate specificity and is capable of degrading bioactive peptides such as incretins involved in blood glucose regulation (1). The results of this study are expected to provide important clues to elucidate the molecular mechanism of the relationship between periodontal disease and systemic diseases, including diabetes.
It has been reported that periodontal disease is associated with various systemic diseases such as diabetes, dementia, and kidney disease. However, but the molecular mechanism of the association has not been clarified yet. The Nemoto group has been studying periodontal peptidases based on the hypothesis that these peptidases are involved in the periodontal-diabetes linkage (2). In the present study, they investigated the degradation of incretins (GLP-1 and GIP) by four DPPs, DPP4, DPP5, DPP7, and DPP11, and found that DPP7 degraded incretins more potently than DPP4. They also clarified the molecular mechanism of the broad substrate specificity of DPP7.
This research was published in the Journal of Biological Chemistry on January 12, 2022.
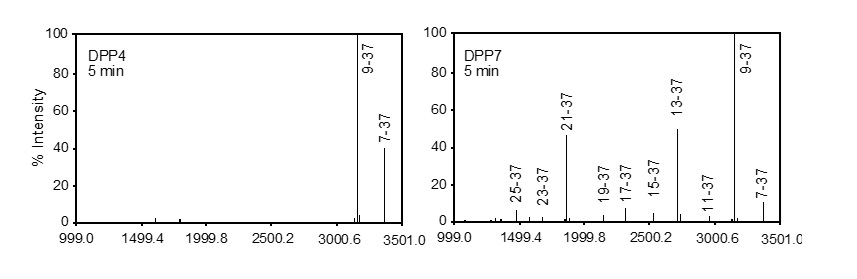 (Fig. 1) P. gingivalis DPP7 degrades incretin GLP-1 more efficiently and thoroughly than DPP4. |
P. gingivalis, a major periodontal bacterium, metabolizes amino acids to produce the energy and all bacterial cell components for its growth. Therefore, the prevention of periodontal disease needs to elucidate the amino acid metabolism in the bacterium.
Bacterial nutritional proteins are degraded into peptides, which pass through the cell wall and into the periplasmic space. In that space, peptides are degraded into dipeptides consisting of two amino acids by four dipeptidyl peptidases (DPP4, DPP5, DPP7, and DPP11) and two exopeptidases (PTP-A and AOP) that assist peptide degradation by DPP. It is then taken up into the cell by Pot, a dipeptide transporter (3). DPPs produce many combinations of dipeptides depending on the amino acid at the P1 position (the second amino acid from the amino terminus of the peptide). However, no DPPs have been found that recognize the hydrophilic amino acids threonine, serine, glutamine, asparagine, and histidine. In this study, Nemoto's group revealed that DPP7, which has been considered to prefer hydrophobic amino acids, has a broader substrate specificity, including these five amino acids.
On the other hand, incretins (GLP-1 and GIP), bioactive peptides secreted from the small intestine, stimulate insulin secretion from the pancreas and regulate blood glucose. These peptides are rapidly degraded by DPP4 in vivo and lose their activity within a few minutes. In this study, MALDI-TOF MS analysis revealed that DPP7 from periodontal bacteria degrades incretins more rapidly than DPP4 (Fig. 1). The involvement of DPP7 in the glucose regulation system was also demonstrated in the glucose tolerance test in mice, where intravenous DPP7 decreased plasma GLP-1 and insulin concentrations, accompanied by an increase in the maximum blood glucose level and prolongation of hyperglycemia.
To elucidate the mechanism of the broad substrate specificity of DPP7, they established a new analytical method containing a fluorescent peptide-substrate and an additional exopeptidase, a two step-exopeptidase analysis. By this method, enzymatic parameters were determined. They found that kcat increased significantly by the presence of the prime side (P1' and P2') residues and that DPP7 can cleave hydrophilic amino acids, which were previously considered to be uncleavable. This present study revealed that P. gingivalis enables to degrade of all polypeptides into dipeptides by the central ability of DPP7 with other DPPs, thus ensuring the growth and virulence of the bacterium
Since the anaerobic oral bacteria inhabiting periodontal pockets contain DPP7 and DPP4 genes and express these peptidase activities (4), it is crucial to determine whether the frequency and amounts of periodontal bacteria transferred from the affected area to the bloodstream increases with the severity of periodontal disease and whether this causes a decrease in blood incretin and insulin levels. Furthermore, periodontal disease may be related to systemic health by degrading incretin, a multifunctional bioactive peptide.
This study is also noteworthy because it enabled quantitative analysis of the effect of C-terminal amino acids on the cleavage position of substrate peptides by proteases, which was previously impossible.
References
(1) Ohara Nemoto, Y., Shimoyama, Y., Ono, T., Sarwar, M.T., Nakasato, M., Sasaki, M., Nemoto, T.K.: Expanded substrate specificity supported by P1’ and P2’ residues enables bacterial dipeptidyl-peptidase 7 to degrade bioactive peptides. J Biol Chem (https://www.jbc.org/article/S0021-9258(22)00025-4/fulltext)
(2) Nemoto, T.K., Ohara-Nemoto, Y.: Dipeptidyl-peptidases: Key enzymes producing entry forms of extracellular proteins in asaccharolytic periodontopathic bacterium Porphyromonas gingivalis. Mol Oral Microbiol (2020) 36(2): 145-145. doi.org/10.1111/omi.12317
(3) Ohara-Nemoto, Y., Sarwar, M.T., Shimoyama Y., Kobayakawa T., Nemoto, T.K.: Preferential dipeptide incorporation of Porphyromonas gingivalis mediated by proton-dependent oligopeptide transporter (Pot). FEMS Microbiol Lett (2020) 367, 1-8. doi: 10.1093/femsle/fnaa204
(4) Ohara-Nemoto, Y., Shimoyma, Y., Nakasato, M., Nishimata, H., Ishikawa, T., Sasaki, M., Kimura, S., Nemoto, T.K.: Distribution of dipeptidyl peptidase (DPP) 4, DPP5, DPP7, and DPP11 in human oral microbiota – potent biomarkers indicating presence of periodontopathic bacteria. FEMS Microbiol Lett (2018) 365(22) doi: 10.1093/femsle/fny221










